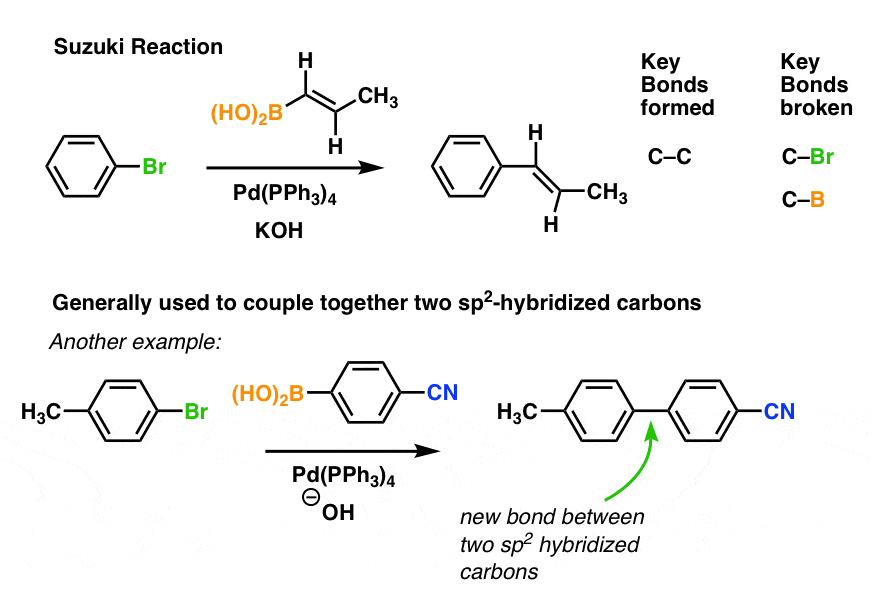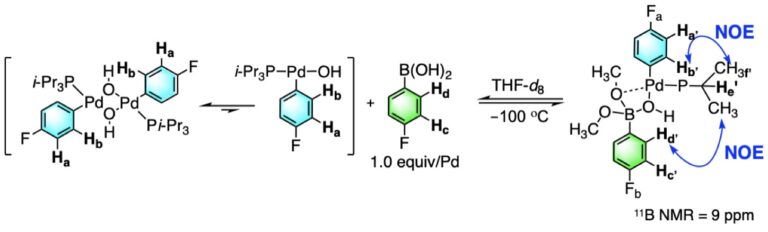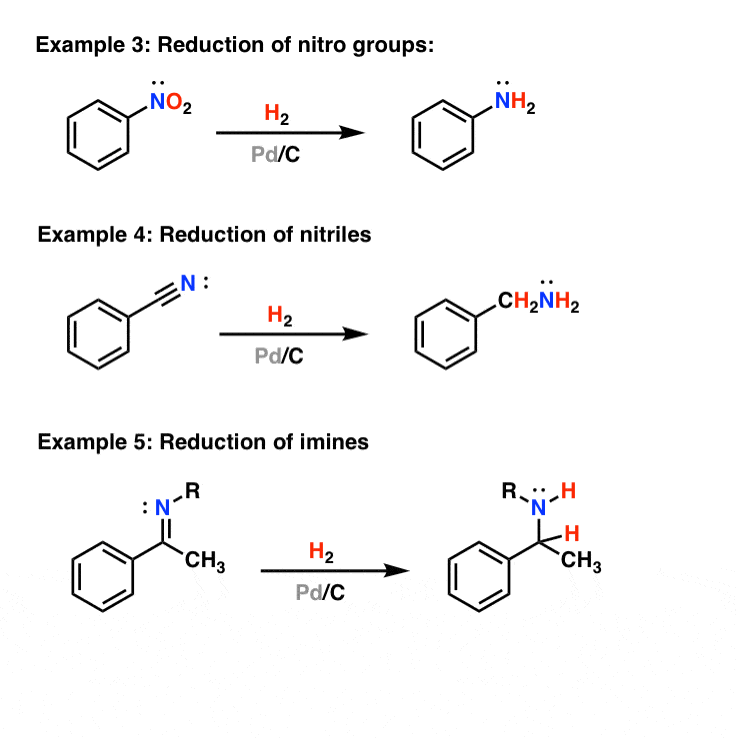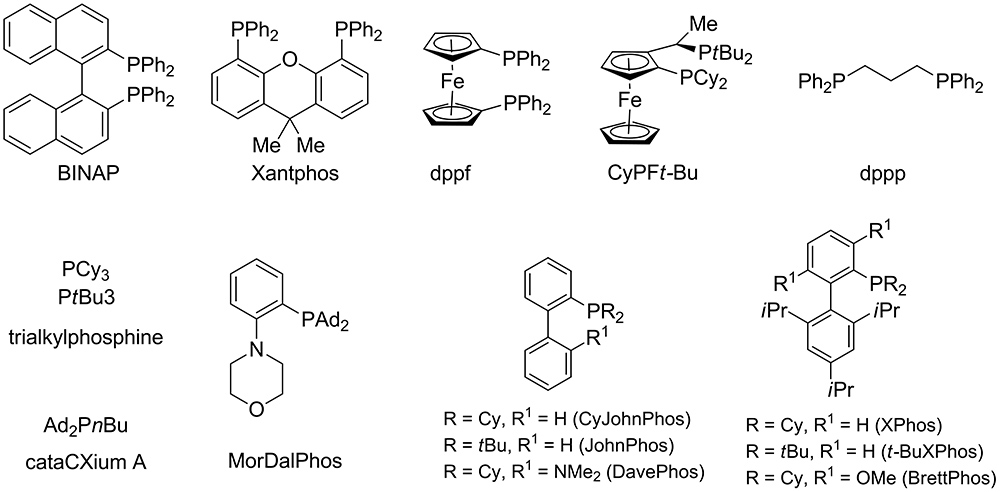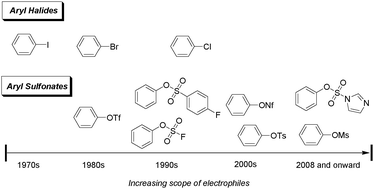
Palladium-catalyzed cross-coupling reactions of aryl mesylates - Chemical Society Reviews (RSC Publishing)

Examples of common palladium-catalyzed reactions in organic synthesis. 24 | Download Scientific Diagram
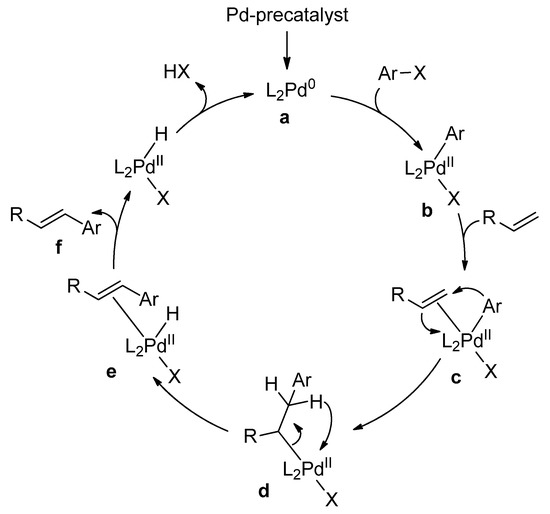
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond

Benzimidazolyl Palladium Complexes as Highly Active and General Bifunctional Catalysts in Sustainable Cross-Coupling Reactions | ACS Catalysis
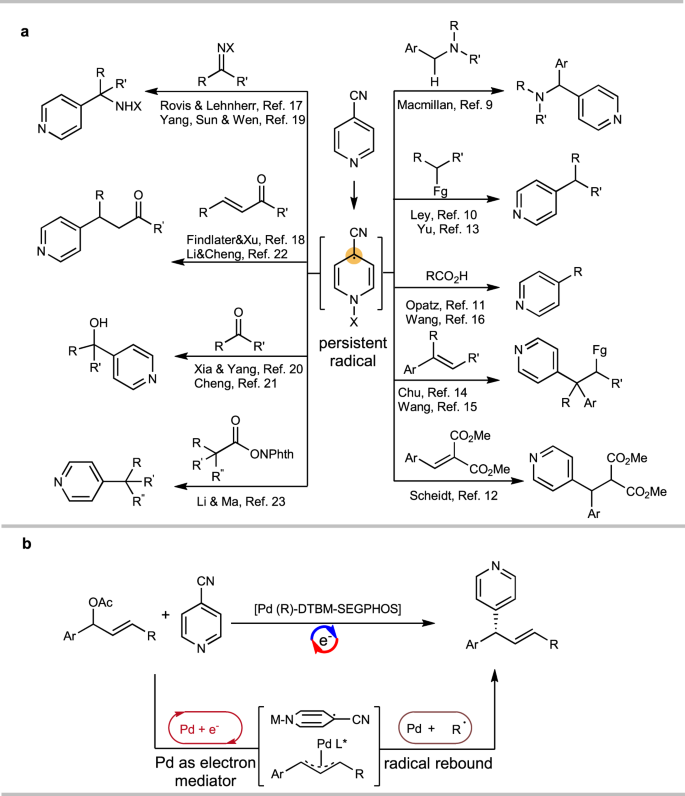
Palladium-catalyzed asymmetric allylic 4-pyridinylation via electroreductive substitution reaction | Nature Communications
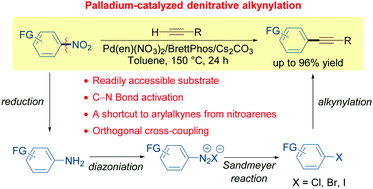
Palladium-catalyzed denitrative Sonogashira-type cross-coupling of nitrobenzenes with terminal alkynes - Chemical Communications (RSC Publishing)
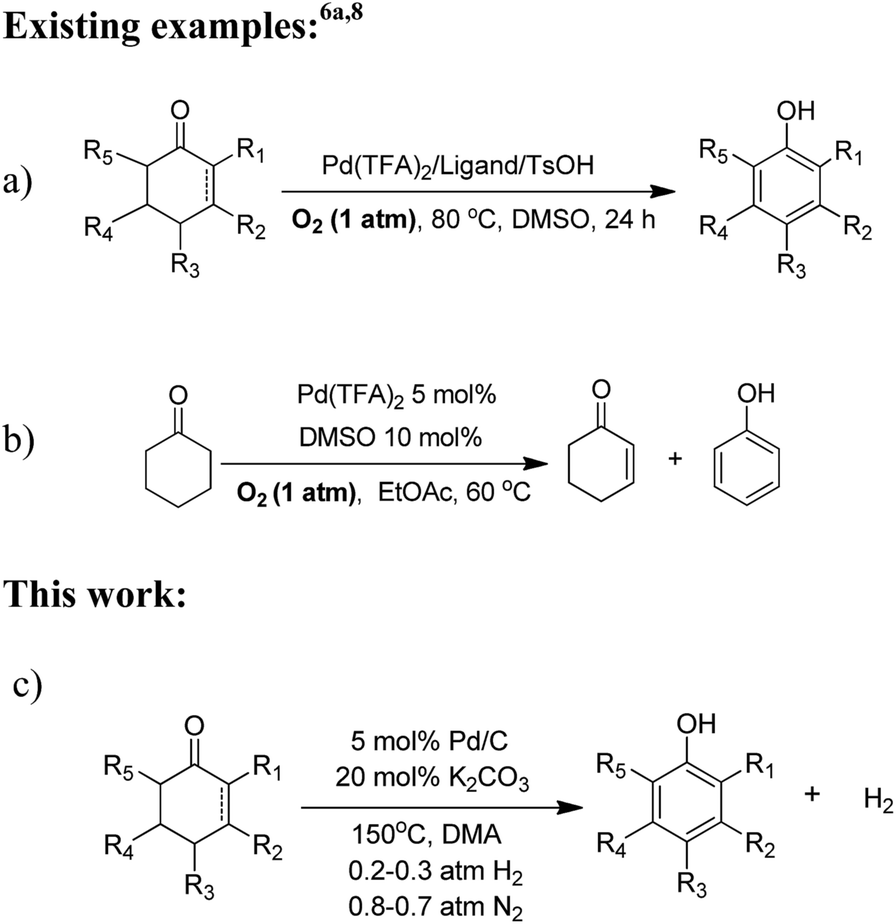
Reaction-activated palladium catalyst for dehydrogenation of substituted cyclohexanones to phenols and H 2 without oxidants and hydrogen acceptors - Chemical Science (RSC Publishing) DOI:10.1039/C5SC01044F

Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO)6 as Carbonyl Source | SpringerLink
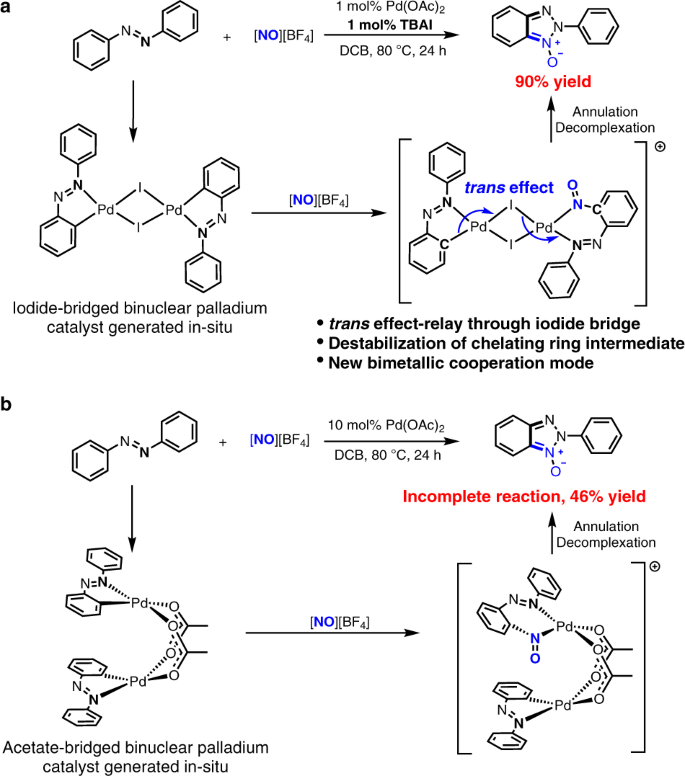
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
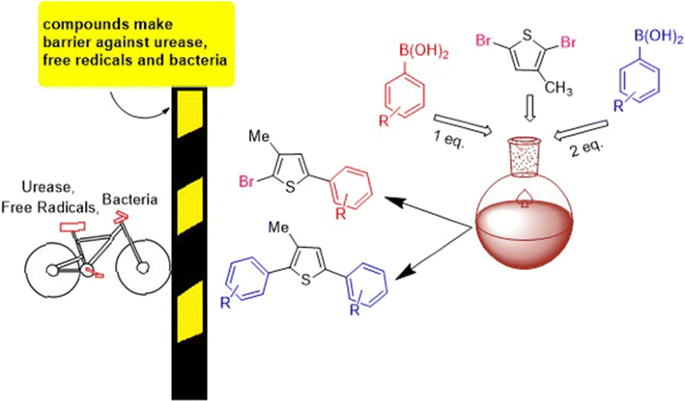
Palladium(0) catalyzed Suzuki cross-coupling reaction of 2,5-dibromo-3-methylthiophene: selectivity, characterization, DFT studies and their biological evaluations | BMC Chemistry | Full Text
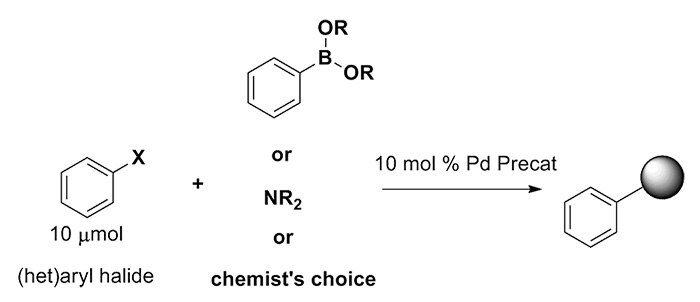
Step-by-Step Guide for KitAlysis High-Throughput Palladium Precatalyst Cross-Coupling Reaction Screening Kit

Palladium nanoparticles catalyzed Suzuki cross-coupling reactions in ambient conditions - ScienceDirect

Palladium(II) phthalocyanines efficiently promote phosphine-free Sonogashira cross-coupling reaction at room temperature - ScienceDirect

Palladium-catalyzed oxidative arene C–H alkenylation reactions involving olefins: Trends in Chemistry